NBS Data Reports

In accordance with Indiana law, the GNBS program tracks and reports data collected while carrying out newborn screening in Indiana.
Screenings help find and identify individuals who do not have any symptoms but may be at risk of having a disease or condition. Diagnostic testing, also called confirmatory testing, is done after screening identifies an individual as possibly having a condition. Testing will confirm or rule out the diagnosis. In other words, screenings identify, and testing confirms.
Initial screens are the first newborn screening collected from a baby. Repeat screens are any additional newborn screenings that are performed on the baby after the initial screen. Screening totals show how many of each type of heel stick screen (initial or repeat) happened each year. Repeat screenings are usually performed if the initial screen is presumptive positive.
Presumptive positive (pre-positive) screens are any screen that resulted potentially positive for a condition. Pre-positive screens require additional testing through a diagnostic test to confirm or rule out the diagnosis. Sometimes the blood collected from the initial screen is not of good enough quality to be analyzed. If this happens, a repeat screen will need to be collected.
Confirmed positive screens include all presumptive positive screens that were confirmed to be positive for the condition through confirmatory testing.
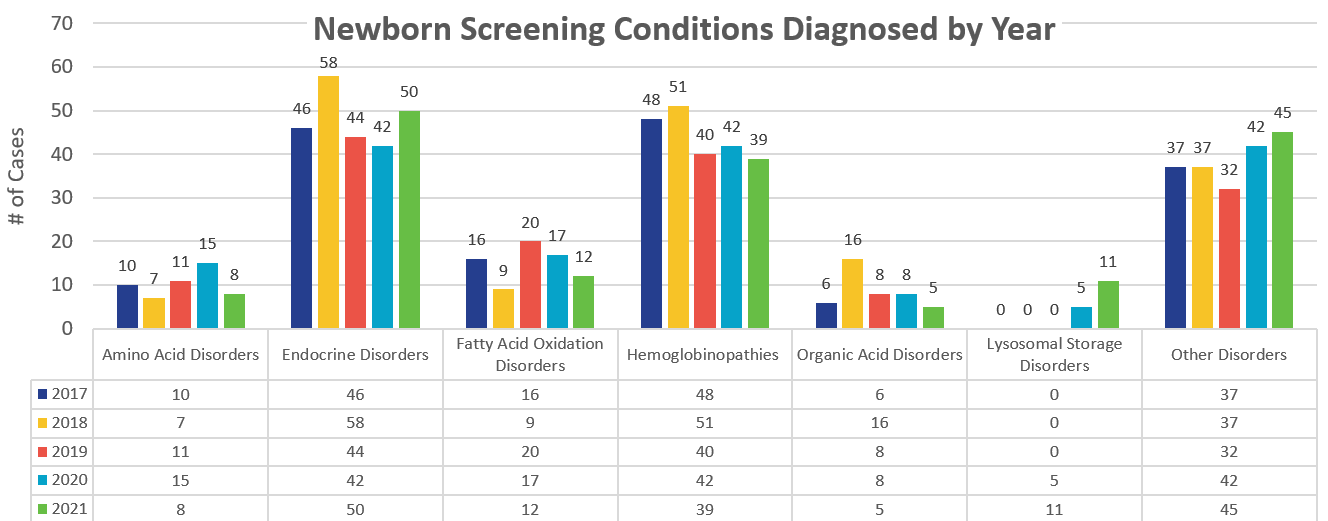
The reports below display the total number of confirmed positive cases of conditions in a given year.
Please note that these totals only reflect the number of cases that were confirmed through confirmatory testing. This does not include presumptive positive cases that did not receive confirmatory testing or were ruled out.
Confirmed NBS Conditions
| 2017 | 2018 | 2019 | 2020 | 2021 | |
|---|---|---|---|---|---|
| Total Confirmed Amino Acid Disorders | 10 | 7 | 11 | 15 | |
| Argininemia (ARG) | 0 | 0 | 0 | 0 | |
| Argininosuccinic Aciduria (ASA) | 1 | 0 | 1 | 0 | |
| Benign Hyperphenylalaninemia (H-PHE) | 0 | 4 | 2 | 5 | |
| Biopterin Defect in Cofactor Regeneration (BIOPT-REG) | 0 | 0 | 0 | 0 | |
| Citrullinemia, Type I (CIT) | 1 | 0 | 0 | 1 | |
| Citrullinemia, Type II (CIT II) | 0 | 0 | 0 | 0 | |
| Classic Phenylketonuria (PKU) | 7 | 2 | 8 | 8 | |
| Hypermethioninemia (MET) | 0 | 0 | 0 | 0 | |
| Homocystinuria (HCY) | 1 | 0 | 0 | 0 | |
| Maple Syrup Urine Disease (MSUD) | 0 | 0 | 0 | 0 | |
| Tyrosinemia, Type I (TYR I) | 0 | 1 | 0 | 1 | |
| Tyrosinemia, Type II (TYR II) | 0 | 0 | 0 | 0 | |
| Tyrosinemi, Type III (TYR III) | 0 | 0 | 0 | 0 | |
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Confirmed Endocrine Disorders | 46 | 58 | 44 | 42 | |
| Congenital Adrenal Hyperplasia (CAH) | 3 | 7 | 2 | 2 | |
| Congenital Hypothyroidism (CH) | 43 | 51 | 42 | 40 | |
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Confirmed Fatty Acid Oxidation Disorders | 16 | 9 | 20 | 17 | |
| 2, 4 Dienoyl-CoA Reductase Deficiency (DE RED) | 0 | 0 | 0 | 0 | |
| Carnitine Acylcarnitine Translocase Deficiency (CACT) | 0 | 0 | 0 | 0 | |
| Carnitine Palmitoyltransferase Type I Deficiency (CPT-I) | 0 | 0 | 0 | 0 | |
| Carnitine Palmitoyltransferase Type II Deficiency (CPT-II) | 0 | 0 | 0 | 1 | |
| Carnitine Uptake Defect (CUD) aka Carnitine Transporter Deficiency (CTD) | 1 | 0 | 0 | 0 | |
| Glutaric Acidemia, Type II (GA-2) | 0 | 2 | 0 | 0 | |
| Long Chain L-3 Hydroxyacyl-CoA Dehydrogenase Deficiency (LCHAD) | 0 | 2 | 0 | 0 | |
| Mediu-Chain Acyl-CoA Dehydrogenase Deficiency (MCAD) | 6 | 5 | 11 | 4 | |
| Medium-Chain Ketoacyl-CoA Thiolase Deficiency (MCAT) | 0 | 0 | 0 | 0 | |
| Medium/Short-ChainL-3 Hydroxyacyl-CoA Dehydrogenase Deficiency (M/SCHAD) | 0 | 0 | 0 | 0 | |
| Short-Chain Acyl-CoA Dehydrogenase Deficiency (SCAD) | 2 | 2 | 4 | 8 | |
| Trifunctional Protein Deficiency (TFP) | 0 | 0 | 0 | 0 | |
Very Long-Chain Acyl-CoA Dehydrogenase Deficiency (VLCAD) | 5 | 0 | 5 | 4 | |
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Confirmed Hemoglobinopathies | 43 | 48 | 37 | 40 | |
| Hb S/S (Sickle Cell Anemia) | 26 | 28 | 26 | 23 | |
| Hb S/C | 12 | 17 | 8 | 15 | |
| Hb S/Variant | 1 | 0 | 0 | 0 | |
| Hb S/Beta Thalassemia | 4 | 3 | 3 | 2 | |
| Beta Thalassemia | 1 | 0 | 0 | 0 | |
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Confirmed Organic Acid Disorders | 6 | 16 | 8 | 8 | |
2-Methyl-3-Hydroxybutyric Acidemia (2M3HBA) | 3 | 8 | 4 | 0 | |
2-Methylbutyrylglycinuria (2MBG) aka 2-Methylbutyryl CoA Dehydrogenase Deficiency | 0 | 3 | 0 | 0 | |
3-Hydroxy-3-Methylglutaric Aciduria (HMG) | 0 | 0 | 0 | 0 | |
3-Methylcrotonyl-CoA Carboxylase Deficiency (3-MCC) | 2 | 4 | 1 | 4 | |
| 0 | 0 | 0 | 0 | ||
Beta-Ketothiolase Deficiency (BKT) | 0 | 0 | 0 | 0 | |
| 0 | 0 | 1 | 0 | ||
| 0 | 0 | 1 | 0 | ||
| 0 | 0 | 0 | 0 | ||
| 0 | 0 | 0 | 0 | ||
| 0 | 0 | 0 | 0 | ||
Methylmalonic Acidemia (Methymalonyl-CoA Mutase Deficiency) (MUT) | 0 | 0 | 1 | 0 | |
| 0 | 0 | 0 | 0 | ||
| 1 | 1 | 0 | 4 | ||
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Confirmed Lysosomal Storage Disorders (screening began 07/01/2020) | - | - | - | 5 | |
| Krabbe | - | - | - | 0 | |
| Mucopolysaccharidosis Type I (MPS-I) | - | - | - | 0 | |
| MPS-I Pseudodeficiency | - | - | - | 2 | |
| Pompe - Infantile Onset | - | - | - | 1 | |
| Pompe - Late Onset | - | - | - | 2 | |
| 2017 | 2018 | 2019 | 2020 | 2021 | |
| Total Other Disorders | 31 | 33 | 30 | 42 | |
| Biotinidase Deficiency (BIOT) | 4 | 7 | 6 | 6 | |
| Classic Galactosemia (GALT) | 2 | 2 | 1 | 1 | |
| Duarte Galactosemia | |||||
| Cystic Fibrosis (CF) | 25 | 24 | 17 | 30 | |
| Severe Combined Immunodeficiency (SCID) - Screening began 07/2018 | - | 0 | 2 | 0 | |
| Spinal Muscular Atrophy (SMA) - Screening began 07/2018 | - | 0 | 4 | 5 | |
| Total Confirmed Newborn Screening Conditions | 140 | 152 | 141 | 154 |
Click on image to expand.
NBS Condition Carrier Data
| NBS Condition Trait/Carrier Type | 2017 | 2018 | 2019 | 2020 | 2021 |
|---|---|---|---|---|---|
| Cystic Fibrosis Carrier | 243 | 209 | 204 | 194 | |
| Hemoglobin S Trait (Sickle Cell Carrier) | 1022 | 1023 | 1185 | 1208 |
Newborn Screen Type: Initial vs. Repeat
| Year | Total Screens | Initial Screens | Repeat Screens | % Repeats |
|---|---|---|---|---|
| 2021 | ||||
| 2020 | 88899 | 78806 | 10093 | 11.4% |
| 2019 | 91160 | 81385 | 9775 | 10.7% |
| 2018 | 94676 | 82015 | 12661 | 13.4% |