Electronic Lab Reporting for Communicable Diseases
Electronic Laboratory Reporting (ELR) is the automated reporting of communicable diseases to the Indiana Department of Health (IDOH), in accordance with Indiana’s Communicable Disease Rule (410 IAC) and Indiana’s Reportable Diseases and Results/Pathogens. The benefits of ELR are significant for both healthcare providers and public health authorities, as it improves timeliness of reporting and reduces manual errors. This efficiency allows them to take appropriate actions to limit the spread of diseases and manage impacts effectively. Specifically,
- ELR uses standardized messaging formats (HL7), coding systems (LOINC) and terminology (SNOMED), facilitating seamless data exchange across different healthcare jurisdictions and systems.
- Implementing ELR helps facilities meet state and federal reporting mandates, including the CMS Promoting Interoperability Programs (PI).
ELR Submission Method
Electronic submission is the secure and automatic transmission of laboratory results from participating facilities to IDOH for communicable disease investigation. To ensure the onboarding process runs efficiently, facilities should engage in the following preliminary preparation:
- Establish credentials through Access Indiana. Register for Promoting Interoperability
- Prepare a list which includes your organization's NPI, CLIA, and location information
ELR Onboarding Process
The ELR Onboarding guide (HL7 Checklist) contains information about all required data elements, formatting of HL7 messages, resources, and best practices for successful onboarding and submission to IDOH. This guide serves as a companion to the HL7 Version 2.5.1 Implementation Guide provided by HL7.org.
- IDOH Intake
- Complete the Sample LOINC List Template
- Connectivity
- Secure File Transfer Protocol (SFTP)
- Testing in IDOH Test Environment
- Generate a single test message into IDOH test environment to check connectivity and message structure
- When all test messages have fewer than 10 errors, send messages for the LOINC code selected for testing
- Validation
- Validations are conducted in the IDOH system known as National Electronic Disease Surveillance System (NEDSS) Base System (NBS).
- When IDOH validation is complete, you will receive approval to start sending ELR messages to production.
- Types of validations
- Structural validation
- Review the structure of ELR messages to ensure HL7 standards are met
- Data quality validation
- Assess the data accuracy, completeness, timeliness, and jurisdictional routing
- Other validation
- Assess and resolve issues identified by IDOH during the validation process
- Structural validation
- Types of validations
- Production Approval
- Receive approval confirmation email from IDOH
- IDOH will place the participant into the NBS production environment
- The participant will discontinue previous methods of reporting Indiana reportable pathogens/results
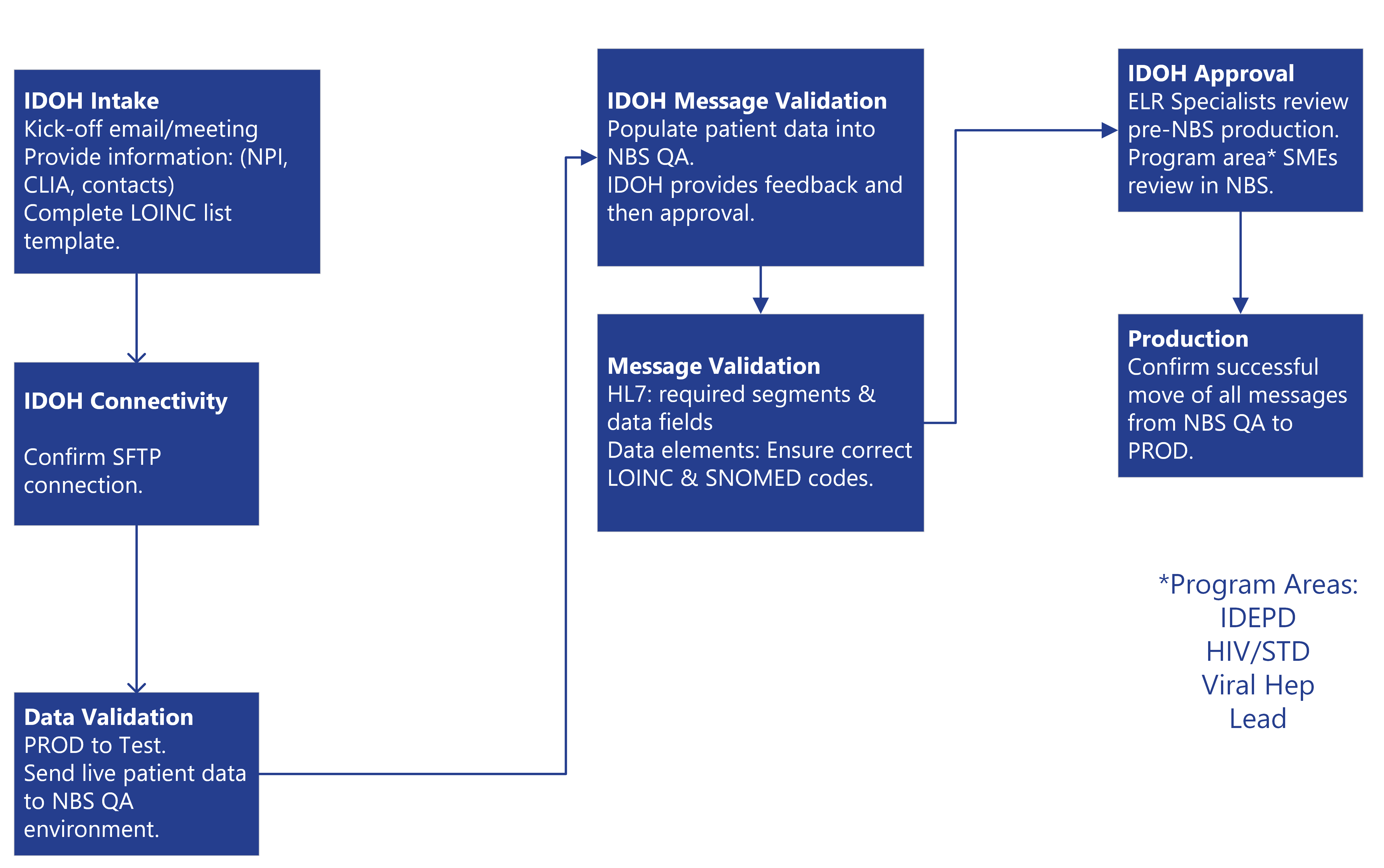
ELR Onboarding Process Diagram
The diagram below provides a visual representation of the onboarding steps described above.
Page last reviewed/updated: April 2026

